Authors: Neelam Jain, Prashant M Onkar, Sunitha Pradeep, Devarajan P, Anjali Gupta, Lalit K Sharma, Shilpa R Satarkar, Renu Sharma, Akanksha Baghel, Bavaharan R, Rijo M Choorakuttil, Praveen K Nirmalan for the Samrakshan Team
Author Affiliations:
- Neelam Jain, Jain Ultrasound centre, C-112, B block, Dispensary road Sonari, Jamshedpur Jharkhand, India
- Prashant M Onkar, Associate Professor, Department of Radiodiagnosis, NKPSIMS, Nagpur, Maharashtra, India
- Sunitha Pradeep, Associate Professor, Department of Radiodiagnosis, JIPMER, Puducherry, India
- Devarajan P, Nethra Scans and Genetic Clinic, Tiruppur, Tamil Nadu, India
- Anjali Gupta, Anjali Ultrasound and Colour Doppler centre, 2nd floor, Shanti Madhuban Plaza, Delhi Gate, Agra, Uttar Pradesh, India,
- Lalit K Sharma, Raj Sonography & X- Ray Clinic, Baiju Choraha, Nayapura, Guna, Madhya Pradesh, India
- Shilpa R Satarkar, Antarang Sonography and Colour Doppler Center, Satarkar Hospital, Plot 20. Tilaknagar, Aurangabad, Maharashtra, India
- Renu Sharma, Dr Renu’s Diagnostic Center,E 6 Basant Vihar, Sikar, Rajasthan, India
- Akanksha Baghel, Baghel Sonography Center. Front of Janpat Office, near District Hospital, Harda, Madhya Pradesh, India
- Bavaharan R, Fetocare Magnum Imaging and Diagnostics, Trichy, Tamil Nadu, India
- Rijo M Choorakuttil, National Coordinator for Samrakshan IRIA, AMMA Center for Diagnosis and Preventive Medicine, Kochi, Kerala, India
- Praveen K Nirmalan, Chief Research Mentor, AMMA ERF, AMMA Center for Diagnosis and Preventive Medicine, Kochi, Kerala, India
Running Title: 2nd Trimester screening
Corresponding Author: Rijo M Choorakuttil, National Coordinator for Samrakshan IRIA, AMMA Center for Diagnosis and Preventive Medicine, Kochi, Kerala, India. E-mail: rijomc@gmail.com
Keywords: Uterine artery PI, Preterm Pre-eclampsia, Fetal Growth Restriction, 2nd trimester screening, Samrakshan, India
Abstract
Aim: To describe the incidence and associations of early fetal growth restriction and high risk for preterm PE in pregnant women screened using the 2nd trimester protocol of Samrakshan, India
Methods: The 2nd trimester protocol of Samrakshan involves collection of clinico-demographic details, mean arterial pressure, fetal biometry to estimate fetal weight and abdominal circumference, measurement of placental thickness and amniotic fluid, uterine artery Doppler study and a TIFFA study. A gestational age specific mean uterine Artery PI >95th percentile was considered as abnormal. A competing risk Bayesian model-based algorithm was used to determine the risks for development of preterm pre-eclampsia. Early Fetal Growth Restriction was defined as an EFW <3rd percentile irrespective of Doppler status or an EFW 3-10th percentile with abnormal mean uterine artery PI Doppler study.
Results: The records of 421 pregnant women screened as part of the Samrakshan program in 16 districts of 8 states of India from July 2019 to February 2020 were analyzed. Twenty-six (6.18%, 95% CI: 4.25, 8.90) women were identified as at risk for preterm PE and 13 (3.09%, 95% CI: 1.81, 5.21) fetuses had early fetal growth restriction. Twenty-nine fetuses (6.89%, 4.84%-9.72) were at high risk for fetal growth restriction and 26 (6.18%, 95% CI: 4.25, 8.90) fetuses were SGA fetuses. The incidence of FGR showed a significant reduction ( p<0.001) if we integrate Doppler with EFW<10th percentile as the parameters defining FGR. Fetal AC and EFW >90th percentile and mean uterine artery PI >95th percentile was associated with higher risk for preterm PE and early FGR.
Conclusion: Incorporating Doppler studies of the uterine artery in the 2nd trimester screening protocol led to a significant decrease of fetuses identified as early FGR and change in risk categorization of fetuses.
Introduction:
Samrakshan is a national program of the Indian Radiological and Imaging Association (IRIA) to reduce perinatal mortality in India.[1] Samrakshan focuses on pre-eclampsia (PE) and fetal growth restriction (FGR) and utilizes trimester specific protocols integrating colour Doppler ultrasound studies and competing risk algorithms to identify pregnant women at risk for preterm PE and FGR.[1] We have previously described the results of integrating colour Doppler Studies with routine 3rd trimester exams and its impact on the diagnosis of FGR.[2,3] In this manuscript, we describe the results of integrating colour Doppler Studies with routine 2nd trimester exams of pregnant women.
Methods
Each woman screened as part of Samrakshan was assigned a unique identification number (ID) at the first screening visit, which is carried throughout the course of pregnancy. The unique ID helps to link records with previous visits. In the 2nd trimester, clinico-demographic details including age of the woman, parity, type of conception, maternal smoking, previous risk of preeclampsia, date of previous delivery and prior birth (if applicable), use of low dose aspirin, and medical co-morbidity were collected. The systolic and diastolic blood pressure of both arms were measured twice in the sitting position and a mean arterial pressure (MAP) was determined for each woman.[4] A Targeted Imaging for Fetal Anomalies (TIFFA) scan was done between 19 and 22 weeks to identify structural abnormalities. Placental thickness was assessed as the vertical length measurement of placenta excluding retroplacental myometrium. The volume of liquor was determined based on the single deepest vertical pocket and a single deepest vertical pocket between 2-8 cm was considered as normal. [5,6] The estimated fetal weight (EFW) of each fetus was calculated from the head circumference (HC), abdominal circumference (AC) and the femoral diaphyseal length (FL) using the Hadlock III formula. The fetal AC and EFW of each fetus was categorized as <3rd, 3-10th and >10th percentiles.[7]
The uterine artery Doppler study was performed through a transabdominal approach. Pulsed‐wave Doppler was used to obtain the spectral waveforms after identifying the right and left uterine arteries at the cross over with the external iliac arteries and the Pulsatility Index (PI) was measured when at least three similar consecutive waveforms were obtained. A mean uterine artery Doppler PI >95th percentile was considered as an abnormal study.[8]
The competing risk Bayesian model-based algorithm of the Fetal Medicine Foundation, available as an online calculator at https://fetalmedicine.org/research/assess/preeclampsia/first-trimester was used to determine the risks for development of preterm pre-eclampsia. Early Fetal Growth Restriction was defined as an EFW <3rd percentile irrespective of Doppler status or an EFW 3-10th percentile with abnormal mean uterine artery PI Doppler study. A fetus with EFW 3-10th percentile with normal Doppler study was defined as a small for gestational age (SGA) baby. A fetus with an EFW >10th percentile but with an abnormal mean uterine artery PI was defined as at risk for development of FGR. A fetus with an EFW >10th percentile and normal Doppler study was considered as appropriate for gestational age (AGA) and at lower risk.
Data was entered online and stored in de-identified form in a password protected database in Samrakshan. Point estimates and 95% confidence intervals around point estimates were determined using the Open Epi program. A binomial test was used to determine significant differences in proportions and a pairwise correlation test was used to determine correlation of fetal AC and EFW. Potential differences in MAP and placental thickness by risk for development of preterm PE and early FGR was assessed using a student’s t-test. The 75th, 80th and 90th percentiles of MAP were determined and the area under receiver operator characteristic curve (AUROC) was determine determined for these measures of MAP with the risk of development of preterm PE. The diagnostic accuracy, false positive rate (FPR), false negative rate (FNR) and positive likelihood ratios were determined for the 75th, 80th and 90th percentile of MAP with the risk for development of preterm PE. A bivariate analysis was used to explore potential associations with early FGR and risks for development of preterm PE and expressed as odds ratios and 95% confidence intervals. A p value less than 0.05 was considered as statistically significant.
Results
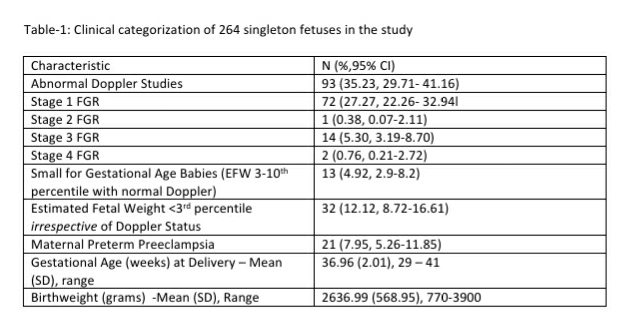
The records of 421 pregnant women screened as part of the Samrakshan program in 16 districts of 8 states of India from July 2019 to February 2020 were analyzed. The majority of women screened (see Table-1) were nulliparous (n=294, 69.83%) and aged between 20 and 29 years (n=287, 68.10%). Fetal abdominal circumference percentiles had a good correlation with estimated fetal weight percentiles (correlation coefficient 0.77, 95% CI 0.72, 0.80).
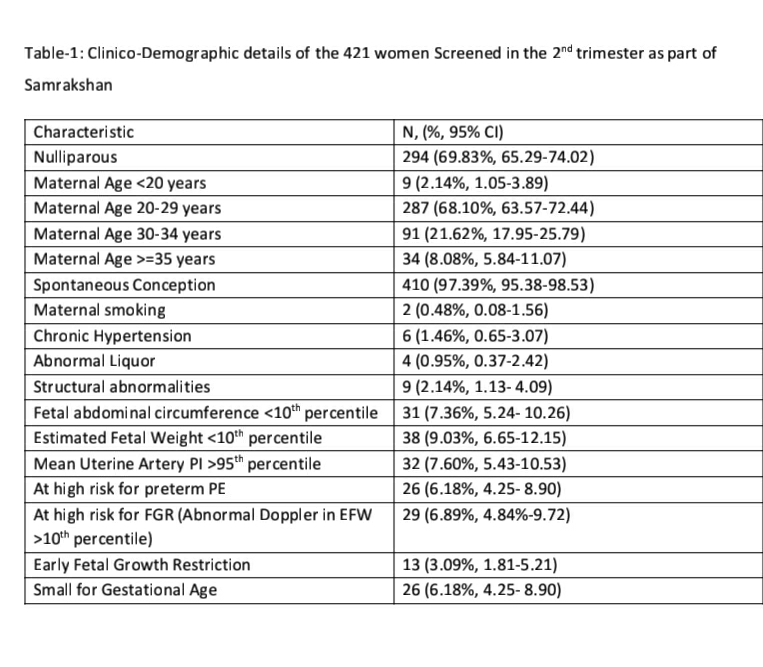
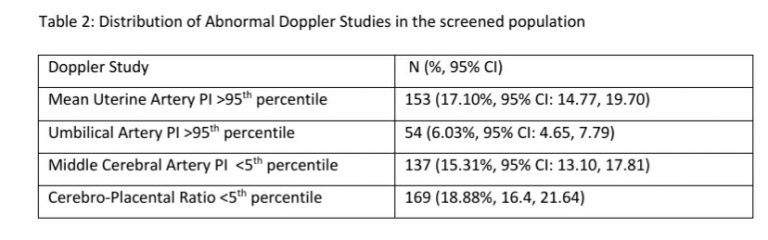
Twenty-six (6.18%, 95% CI: 4.25, 8.90) women were identified as at risk for preterm PE and 13 (3.09%, 95% CI: 1.81, 5.21) women had fetuses with early fetal growth restriction. Only 3 (23.08%, 95% CI: 8.18, 50.25) of the 13 fetuses with early FGR had an abnormal mean uterine artery PI. Twenty-nine fetuses (6.89%, 4.84%-9.72) had an EFW >10th percentile with a mean uterine artery PI >95th percentile and were considered at high risk for the development of fetal growth restriction. Twenty-six (6.18%, 95% CI: 4.25, 8.90) fetuses had an EFW <10th percentile with a normal mean uterine artery PI and were considered as SGA fetuses.
The incidence of early FGR (n= 42, 9.98%, 95% CI: 7.47, 13.21) if we defined FGR as the presence of any one of EFW or AC <10th percentile did not differ significantly (p=0.64) if we defined FGR based only on an EFW <10th percentile (n=38, 9.03%, 95% CI: 6.65, 12.15). However, the incidence of FGR showed a significant reduction (p<0.001) if we integrate Doppler with EFW<10th percentile as the parameters defining FGR.
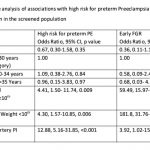
In a bivariate analysis, high risk for preterm PE and early FGR was not associated with either parity or maternal age in the screened population (see Table-2). Fetal AC and EFW <10th centile was significantly associated with high risk for preterm PE and early FGR but the magnitude of association was much higher for early FGR. Mean Uterine artery PI >95th percentile was significantly associated with both high risk for preterm PE and early FGR but the magnitude of association was much higher for high risk for preterm PE.
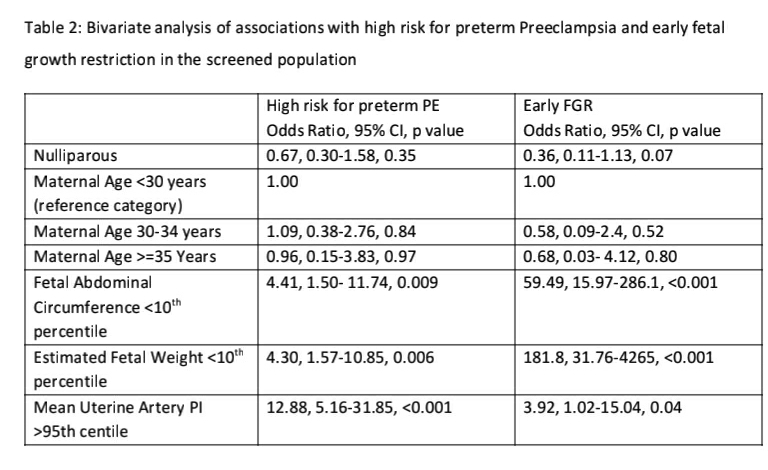
Bivariate analysis was not attempted for maternal smoking, chronic hypertension or abnormal liquor given the small number of cases in each category. Placental thickness did not differ significantly among those at high risk for preterm PE and those at low risk for preterm PE (p=0.66) or those with early FGR and those without FGR (p=0.33).
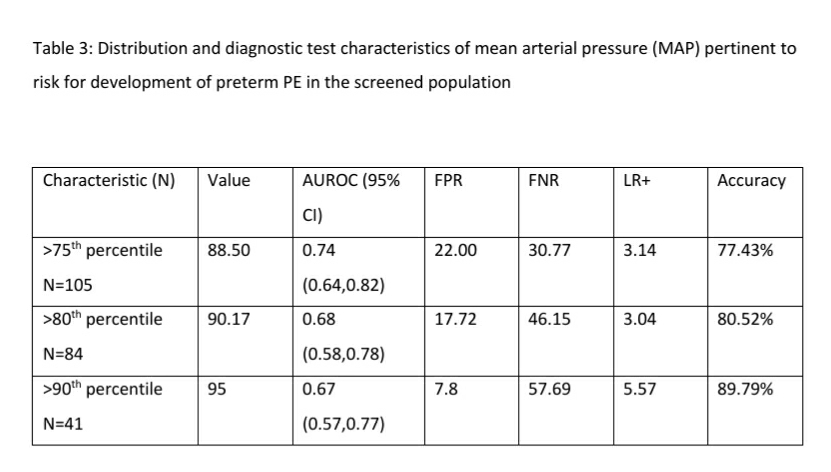
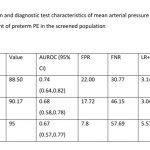
The diagnostic test characteristics of MAP with the risk for development of preterm PE is shown in Table 3. A MAP >90th percentile (MAP >95) had a diagnostic accuracy of 89.79% and a positive likelihood ratio (LR +) of 5.57 for a false positive rate of 7.8%. A MAP >95th percentile had a LR+ of 7.56 and a diagnostic accuracy of 92.16 for a false positive rate of 3.54. However, the false negative rate of MAP >95th percentile was much higher (73.08%) compared to the >90th percentile of MAP (57.69%)
Discussion
The predictive performance of uterine artery Doppler is better for early‐onset PE and Uterine artery Doppler velocimetry tends to predict the more severe and complicated cases. A previous study of more than 32,000 pregnant women reported that uterine artery PI alone can predict as high as 85% of preterm PE at a false‐positive rate of 10% in the 2nd trimester.[9] The risk for early PE increases with increasing uterine artery resistance. [9] Bilateral diastolic notching in the uterine artery Doppler waveform is also associated with increased risk for PE but uterine artery PI is associated with better sensitivity than notching for the same false positive rate, rendering it unnecessary to add identification of notching to the screening protocol. [10,11]
A previous study reported that a second‐trimester model using uterine artery PI, maternal factors (including BMI, ethnic origin, previous obstetric history, smoking status, type of conception, medical history) and mean arterial blood pressure can detect as many as 100% of women who develop early PE for a false‐positive rate of 10%. [12] The sensitivity for late PE and gestational hypertension was 56.4% and 54.1%, respectively. [12] The integration of mean uterine artery Doppler PI and the Bayesian risk algorithm identified 6.18% of the 2nd trimester population screened in Samrakshan as at high risk for the development of preterm PE. These estimates of possible incidence are consistent with the known estimates of pre-eclampsia in India (5-8%). The population is being followed up to determine the diagnostic effectiveness of the 2nd trimester protocol aligned with perinatal outcomes. Parity, maternal age, maternal co-morbidity, prior obstetric history, were positive for only 2.37% of the screened population indicating their inadequacy, in isolation, to identify those at risk for preterm PE.
The results of this study suggest that MAP >90th percentile may indicate a higher risk for the development of preterm PE. The MAP >95th percentile gives a higher LR+ and diagnostic accuracy but comes with a much higher false negative rate. Considering the clinical implications of preterm PE, the MAP >90th percentile gives a better balance between LR+ rates, FPR, FNR and diagnostic accuracy. However, we recommend caution while interpreting this result and do not recommend clinical translation at this stage. A larger study with a larger sample size from diverse settings is needed to determine the percentile distribution of MAP. Additionally, the current analysis presents the diagnostic test characteristics of MAP pertinent to the risk of development of preterm PE. We need to further analyse the percentile distribution of MAP with the actual development of preterm PE to obtain more meaningful results that can translate to clinical application.
Fetal AC and EFW showed a good correlation indicating that AC could be used instead of EFW in the diagnosis of FGR. The integration of Doppler in the diagnostic parameters led to a significant reduction in the incidence of fetuses classified as early FGR (from 9.03% to 3.09%). The integration of Doppler also led to a significant reclassification of fetuses at risk. Twenty-six (6.18%, 95% CI: 4.25, 8.90) fetuses with an EFW <10th percentile (would have previously been classified as FGR) had a normal mean uterine artery PI and were now classified as SGA fetuses with a risk of perinatal outcomes similar to AGA fetuses. Twenty-nine fetuses (6.89%, 4.84%-9.72) with an EFW >10th percentile (AGA fetuses) had a mean uterine artery PI >95th percentile and were now classified as at higher risk for the development of fetal growth restriction.
What does this reclassification mean for antenatal care and surveillance? We found the absolute number of fetuses that need closer surveillance did not differ. 42 fetuses need closer surveillance if we considered FGR as an EFW or fetal AC <10th percentile. If we considered the Doppler findings, 13 early FGR and 29 AGA fetuses with abnormal Doppler (total n=42) need closer surveillance. Thus, the workload does not change in terms of absolute numbers.
However, the integration of Doppler results in a shift of a group of fetuses earlier considered as high risk to a lower risk category (SGA) and shifts a group of AGA fetuses previously considered as low risk to a higher risk group. The shift of risk categorization in AGA fetuses is important as they are less closely monitored due to their low risk status. Additionally, the integration of colour Doppler studies helped to differentiate 23% of fetuses with early FGR as having a possible uteroplacental causation. The real implications of this reclassification, however, has to be assessed in terms of perinatal outcomes and the screened population is being followed up to obtain those results.
Our findings suggest that reliance on growth alone or changes in growth alone to identify fetal growth restriction may not be optimal. This finding assumes greater importance when we consider the inherent inaccuracies and variations associated with the ultrasound estimation of growth parameters and their plotting on growth charts. This is an area that needs further study.
The number of fetuses identified as needing closer surveillance due to early FGR and a risk of development of preterm PE and early FGR was significantly higher (p<0.001) than the number of fetuses identified with structural abnormalities (n=9) in the screened population. This finding has implications on the current focus of 2nd trimester screening that is heavily oriented towards the identification of fetal abnormalities.
There was a significant difference in the magnitude of association of fetal AC, EFW and mean uterine artery PI with risk for preterm PE and early FGR. Consistent with global literature and current knowledge of pathophysiology of preterm PE and early FGR, fetal AC and EFW had a significantly higher magnitude of association with early FGR and uterine artery Doppler had a significantly higher magnitude of association with preterm PE.
The National Family Health Survey (NFHS)-4 reported a perinatal mortality rate of 36 per 1000 pregnancies in India.[13] The NFHS-4 reported a neonatal mortality rate of 30 per 1000 live births and a still birth rate of 0.7. [13] Prematurity and low birth weight were identified as the major causes for neonatal mortality in India. [14] Second‐trimester prediction of PE is at least as sensitive as prediction in the first trimester, but has limited value due to the lack of effective interventions that can be offered at this gestational stage.[12, 15] Low dose aspirin started in the first trimester of pregnancy (11-14weeks) appears to reduce the development of PE but is ineffective when started after 20 weeks. [16,17] Although it is too late to prevent the development of PE after second‐trimester prediction, the knowledge can still be useful in guiding follow‐up and management of a pregnancy at risk. [18,19]
The preliminary results of the 2nd trimester screening protocol of Samrakshan indicates that integrating Doppler in the routine 2nd trimester exam may help better with identification of subgroups that need closer surveillance. However, these results have to be validated through further longitudinal studies on larger sample sizes from diverse settings and aligned with perinatal outcomes.
References
- Choorakuttil RM, Patel H, Bavaharan R, Devarajan P, Kanhirat S, Shenoy RS, Tiwari OP, Sodani RK, Sharma LK, Nirmalan PK. Samrakshan: An Indian radiological and imaging association program to reduce perinatal mortality in India. Indian J Radiol Imaging 2019;29:412-7
- Bavaharan R, Choorakuttil RM, Ahuja B, Gupta A, Sharma LK, Baghel A, et al. Routine 3rd Trimester Colour Doppler Ultrasound in Fetuses with Estimated Fetal Weight 10-50th centiles in India- Preliminary Results from the Samrakshan Program. Accessed online from http://fetalradiology.in/2020/01/16/routine-3rd-trimester-colour-doppler-ultrasound-in-fetuses-with-estimated-fetal-weight-10-50th-centiles-in-india-preliminary-results-from-the-samrakshan-program/ on February 1,2020
- Anjali G, Renu S, Sharma LK, Akanksha B, Bavaharan R, Shilpa RS, at al. Colour Doppler studies identify more fetuses at risk for compromise in the third trimester: Preliminary results from Samrakshan India. Accessed online from http://fetalradiology.in/2020/02/09/colour-doppler-studies-identify-more-fetuses-at-risk-for-compromise-in-the-third-trimester-preliminary-results-from-samrakshan-india/ on February 18, 2020
- Poon LC, Zymeri NA, Zamprakou A, Syngelaki A, Nicolaides KH. Protocol for measurement of mean arterial pressure at 11‐13 weeks’ gestation. Fetal Diagn Ther. 2012; 31: 42– 48.
- Williams K. Amniotic fluid assessment. Obstet Gynecol Surv 1993; 48: 795– 800.
- Moise KJ, Jr. Toward consistent terminology: assessment and reporting of amniotic fluid volume. Semin Perinatol 2013; 37: 370– 374.
- Hadlock FP, Harrist RB, Sharman RS, Deter RL, Park SK. Estimation of fetal weight with the use of head, body, and femur measurements: a prospective study. Am J Obstet Gynecol. 1985;151:333–7.
- Sotiriadis A, Hernandez‐Andrade E, da Silva Costa F, et al. ISUOG Practice Guidelines: Role of ultrasound in screening for and follow‐up of pre‐eclampsia. Ultrasound Obstet Gynecol. 2019; 53: 7– 22.
- Yu CK, Smith GC, Papageorghiou AT, Cacho AM, Nicolaides KH, Fetal Medicine Foundation Second‐Trimester Screening Group. An integrated model for the prediction of preeclampsia using maternal factors and uterine artery Doppler velocimetry in unselected low‐risk women. Am J Obstet Gynecol 2005; 193: 429–436.
- Papageorghiou AT, Yu CK, Erasmus IE, Cuckle HS, Nicolaides KH. Assessment of risk for the development of pre‐eclampsia by maternal characteristics and uterine artery Doppler. BJOG 2005; 112: 703– 709.
- Espinoza J, Kusanovic JP, Bahado‐Singh R, Gervasi MT, Romero R, Lee W, Vaisbuch E, Mazaki‐Tovi S, Mittal P, Gotsch F, Erez O, Gomez R, Yeo L, Hassan SS. Should bilateral uterine artery notching be used in the risk assessment for preeclampsia, small‐for‐gestational‐age, and gestational hypertension? J Ultrasound Med 2010; 29:1103– 1115.
- Onwudiwe N, Yu CK, Poon LC, Spiliopoulos I, Nicolaides KH. Prediction of pre‐eclampsia by a combination of maternal history, uterine artery Doppler and mean arterial pressure. Ultrasound Obstet Gynecol 2008; 32: 877–883.
- International Institute for Population Sciences (IIPS) and ICF. 2017. National Family Health Survey (NFHS-4) 2015-16: India. Mumbai: IIPS
- Million Death Study Collaborators, Bassani DG, Kumar R, et al. Causes of neonatal and child mortality in India: a nationally representative mortality survey. Lancet. 2010; 376:1853–1860.
- Khalil A, Maiz N, Garcia‐Mandujano R, Penco JM, Nicolaides KH. Longitudinal changes in maternal serum placental growth factor and soluble fms‐like tyrosine kinase‐1 in women at increased risk of pre‐eclampsia. Ultrasound Obstet Gynecol 2016;47: 324– 331.
- Bujold E, Roberge S, Lacasse Y, Bureau M, Audibert F, Marcoux S, Forest JC, Giguere Y. Prevention of preeclampsia and intrauterine growth restriction with aspirin started in early pregnancy: a meta‐analysis. Obstet Gynecol 2010; 116: 402– 414.
- Roberge S, Demers S, Nicolaides KH, Bureau M, Cote S, Bujold E. Prevention of pre‐eclampsia by low‐molecular‐weight heparin in addition to aspirin: a meta‐analysis. Ultrasound Obstet Gynecol 2016; 47: 548– 553.
- Litwinska M, Wright D, Efeturk T, Ceccacci I, Nicolaides KH. Proposed clinical management of pregnancies after combined screening for pre‐eclampsia at 19–24 weeks’ gestation. Ultrasound Obstet Gynecol 2017; 50: 367– 372.
- Litwinska M, Syngelaki A, Wright A, Wright D, Nicolaides KH. Management of pregnancies after combined screening for pre‐eclampsia at 19–24 weeks’ gestation. Ultrasound Obstet Gynecol 2018; 52: 365– 372.